IJMS | Free Full-Text | Methotrexate Provokes Disparate Folate Metabolism Gene Expression and Alternative Splicing in Ex Vivo Monocytes and GM-CSF- and M-CSF-Polarized Macrophages

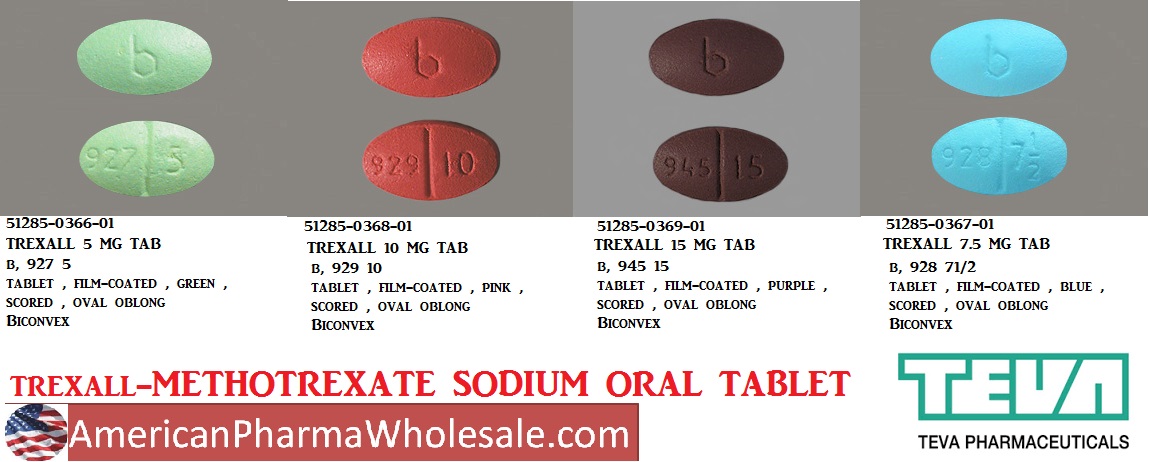
These highlights do not include all the information needed to use TREXALL® safely and effectively. See full prescribing information for TREXALL. TREXALL® (methotrexate tablets), for oral use Initial U.S. Approval: 1953

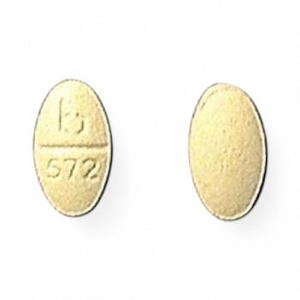
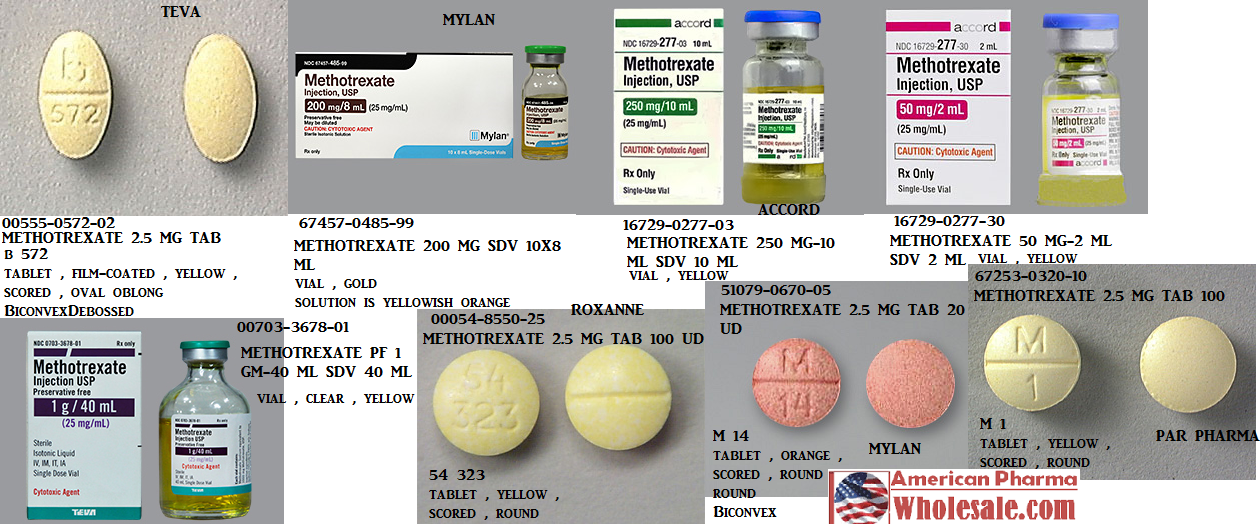
These highlights do not include all the information needed to use METHOTREXATE TABLETS safely and effectively. See full prescribing information for METHOTREXATE TABLETS. METHOTREXATE tablets, for oral use Initial U.S. Approval: 1953

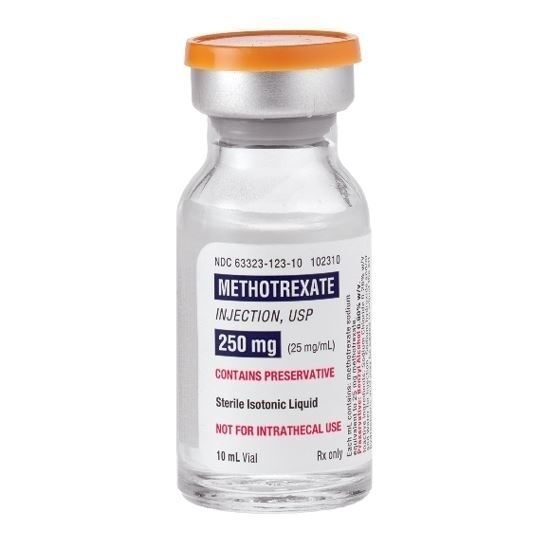
Antineoplastic Agent Methotrexate Sodium, Preservative Free 25 mg / mL Injection Single Dose Vial 10 mL
ChemoClave™ Closed System Transfer Device (CSTD) Surface Wipe Study Using Methotrexate (MTX) as a Marker